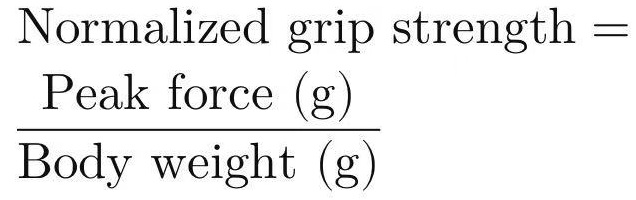Conducting grip strength testing in small animals (mice, rats) requires a standardized protocol to ensure reproducibility and minimize variability. Below is a step-by-step guide based on best practices used in preclinical neuromuscular research.
Select the correct grip plate or grip bar
Forelimb test: Use a mesh grid or a triangular bar attached to the force sensor. The mesh allows the animal to grasp with its forelimb. Hindlimb / all‑limb test: Use a pull bar (often a wire or a small ring) that the animal grasps with both forelimbs, but the force generated includes hindlimb contribution if the animal pushes off.
Calibration
Perform a daily calibration using the manufacturer’s provided weights. Ensure the sensor is zeroed before starting each session.
Angle and stability
Place the grip strength meter on a stable surface. Do angle adjustment, a horizontal plane (0°) is most common for consistent results.
2. Animal Preparation
Acclimatization
Bring animals to the testing room at least 30~60 minutes before starting to reduce stress. This allows them to adjust to differences in ambient temperature, humidity and sounds. Handle them gently for a few days prior to the study to minimize anxiety.
Body weight measurement
Record body weight immediately before the grip strength test. Grip strength is often normalized to body weight (force in grams divided by body weight in grams) to account for size differences.
Avoid sedation
Testing should be performed on awake, freely moving animals. Anesthetics affect neuromuscular function and will confound results.
3. Testing Procedure
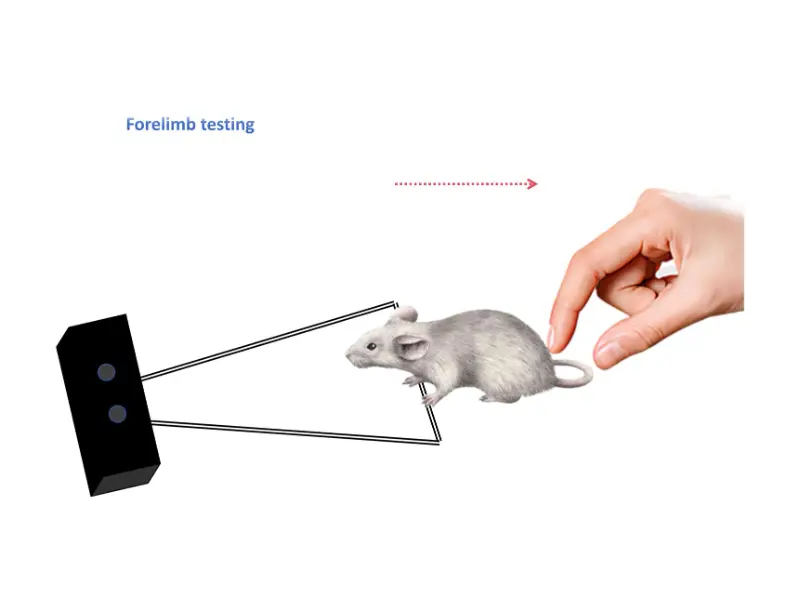
Forelimb Grip Strength
Hold the animal by the base of the tail, allowing its forepaws to grasp the grid or bar.
Gently pull the animal backward along the axis of the sensor at a slow, steady speed (the animal will instinctively resist).
Continue pulling until the animal releases its grip. The meter records the peak force (in grams or Newtons) automatically via a “peak hold” function.
Repeat 3–5 trials per animal with a brief rest (30–60 seconds) between trials to avoid fatigue.
Calculate the average of the trials for that animal.
Hindlimb (or all‑limb) Testing
Use the grid bar.
Place the animal’s forepaws on the bar, then pull horizontally. The animal often uses its hindlimbs to push against a non‑slip surface, generating higher force values.
The same pulling and repetition protocol applies.
4. Data Recording & Normalization
Record the Peak force which is the primary outcome.
Normalize to body weight:
This reduces variability due to differences in animal size.
Record individual trial values, average per animal, and group means ± SEM.
Five consecutive tests were performed on each mouse, and the mean maximum limb muscle strength value (grams) (g) was calculated.
5. Important tips
Pulling speed: Researchers should keep consistent; a slow, steady pull (approx. 1–2 cm/s) reduces startle and variability.
Technician: The same person should perform all tests in a study to avoid inter‑operator variability.
Trial number: Use 3–5 trials; discard the first trial if the animal is clearly uncooperative, but document exclusions.
Fatigue: If force values drop significantly across trials, increase rest time or reduce trial count.
Time of day: Test at the same time of day across groups, as circadian rhythms affect motor performance.
6. Summary
A well‑conducted grip strength test is quick, non‑invasive, and yields highly quantitative data. By standardizing equipment calibration, handling technique, and pulling speed, researchers can obtain reliable, reproducible results that are essential for studies on neuromuscular disease, drug efficacy, and aging. Our Grip Strength Meter equips with high resolution(0.1gf), force calibration and real-time display to make it as the very superb equipment in the quantitative assess neuromuscular function, motor coordination, and muscle strength in small animal for preclinical research.